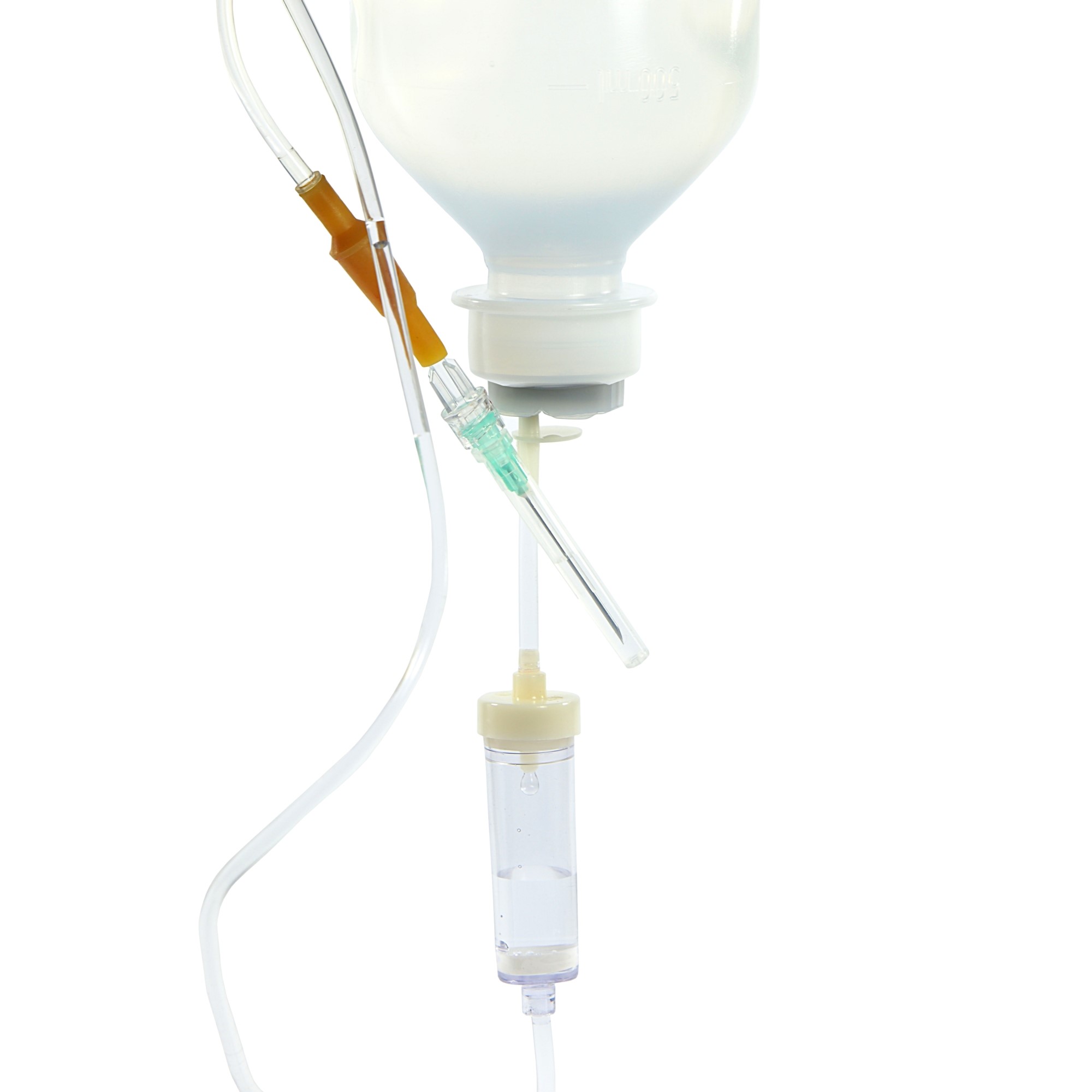
Dear Customer,
Complex application techniques and a wide variety of different and specific application systems are used for infusing medication and nutritional components as well as for the transfusion of blood and blood components in order to administer therapeutic substances to patients both correctly and successfully.
In the early and mid-1600s, many different scientists, including Robert Hooke, Robert Boyle and Christopher Wren, conducted tests with opium infusions on dogs. Wren and his colleague Richard Lower used a quill and pig’s blood for the first blood transfusion on dogs (1665). After several attempts at animal-to-human transfusions – many of which went wrong – blood transfusions were banned for the second time. Once again, this topic disappeared into oblivion for years. It was not until 1795 when the American doctor Philip Syng Physick substantiated human-to-human transfusion. But an outbreak of cholera (1831/32) represented a breakthrough when it was acknowledged that the blood of dehydrated people needed to be treated with lukewarm water and salt. The precursors to IV sets, as they are now known, evolved from 1845 with the development of syringes and needles.
The thermoplastic basic bodies which are identical for all toothbrushes of the same type are simultaneously overmolded with various handle colors in a multi-cavity injection-molding tool. Furthermore, another color or material can be processed during the same manufacturing cycle in order to apply an additional handle component such as a tongue cleaner to the back of the toothbrush head. One essential advantage over sequential production with various overmolding colors is that colors do not need to be changed during the production process. There is also a logistical advantage: if all colors are simultaneously injected in a tool, the 4-colored toothbrushes are directed to the packaging plant right from the conveyor belt. All in all, this saves material, time and storage costs.
However, multi-component technology also poses major challenges for the material. The substrate and overmolding can effectively complement each other. For this, the materials must be compatible. The materials to be selected depend on the function of the injection-molding parts and the respective manufacturing method. As the connection between the process and end product in multi-component injection molding is more complicated than for single-component injection molding, we recommend seeking expert advice when it comes to selecting materials. It is not, for example, only about the compatibility of plastics but also the particular requirements on adhesion. The material developments entail modifications of plastics enabling them to be optimized for the requirements of the actual products to be manufactured. For this reason, ACTEGA DS has adapted the TPE formulae to the special demands on optics, haptics, slip-resistance, secure grip and ergonomics. These SOFT EST. TPE are distinguished by their particular softness, easy coloration properties, good haptics, shorter cycle times in the injection-molding process, particularly good adhesion to PP and PE, and of course compliance with FDA und EU regulations. It is also possible to adapt these good adhesion properties to technical thermoplastics.